Combustion Science Annotations:
Blog Entry 09/12/16:
I believe that when the fire from the candle burned open the plastic balloon. After that happened the hydrogen was released which was set ablaze by the flames from the candle. That was the chemical reaction that we witnessed.
Update:
For a fire to burn it needs an oxidizer, in this case the oxidizers were hydrogen, carbon and even other oxygen atoms leaving the nitrogen to pair up with each other. There is an initial spark of heat that breaks these bonds Every time atoms form a new bond.
Depending on different types of elements you burn can turn the fire into different colors, with the chemical reaction we saw the fire burned bright orange. You can take a look at the video below and see for yourself.
Oxygen has eight protons and eight electrons, so it is always on the hunt for two more electrons to fill the outer shell and make a stable atom.
The chemical reaction is releasing all the new energy that it had gotten and that it could no longer contain.
Another example of this is in a candle. The carbon atoms in the wick bond with the oxygen atom in the air. Or a very slow process of rust: when iron and oxygen combine.
I believe that when the fire from the candle burned open the plastic balloon. After that happened the hydrogen was released which was set ablaze by the flames from the candle. That was the chemical reaction that we witnessed.
Update:
For a fire to burn it needs an oxidizer, in this case the oxidizers were hydrogen, carbon and even other oxygen atoms leaving the nitrogen to pair up with each other. There is an initial spark of heat that breaks these bonds Every time atoms form a new bond.
Depending on different types of elements you burn can turn the fire into different colors, with the chemical reaction we saw the fire burned bright orange. You can take a look at the video below and see for yourself.
Oxygen has eight protons and eight electrons, so it is always on the hunt for two more electrons to fill the outer shell and make a stable atom.
The chemical reaction is releasing all the new energy that it had gotten and that it could no longer contain.
Another example of this is in a candle. The carbon atoms in the wick bond with the oxygen atom in the air. Or a very slow process of rust: when iron and oxygen combine.
Annotations:
"What is light" blog post:
Light is energy, energy has a wavelength. Wavelength is the distance between two peaks in a wave sequence. Electricity can stop the heart, and start it again if it's set at the right frequency.
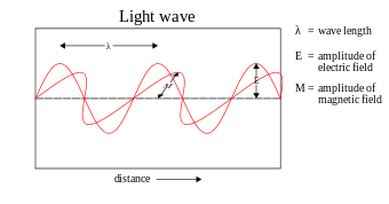
Wavelengths are shown on the electromagnetic spectrum, listed from the longest wavelengths to the shortest. Starting with radio waves, Radio waves are a lot longer/penetrating then light waves, but they are also one of the least harmful. On the other hand, gamma rays have the shortest wavelength and the highest frequency. Gamma rays are be extremely harmful to your body especially when exposed to it for long periods of time. They can completely change your DNA, which I can tell you doesn't come with positive effects. In the middle, there is the visible wavelengths. This is known as the rainbow, and its initials are ROYGBIV.
We can only see different shades of red, orange, yellow, green, blue, indigo, and violet. In class we did an experiment where we burned different elements such as Magnesium, Copper, Lithium, Potassium, Boron, and Sodium. Each element produced its own shade of color from the visible wavelength spectrum.
Wavelengths are shown on the electromagnetic spectrum, listed from the longest wavelengths to the shortest. Starting with radio waves, Radio waves are a lot longer/penetrating then light waves, but they are also one of the least harmful. On the other hand, gamma rays have the shortest wavelength and the highest frequency. Gamma rays are be extremely harmful to your body especially when exposed to it for long periods of time. They can completely change your DNA, which I can tell you doesn't come with positive effects. In the middle, there is the visible wavelengths. This is known as the rainbow, and its initials are ROYGBIV.
We can only see different shades of red, orange, yellow, green, blue, indigo, and violet. In class we did an experiment where we burned different elements such as Magnesium, Copper, Lithium, Potassium, Boron, and Sodium. Each element produced its own shade of color from the visible wavelength spectrum.
Phases Of Change/Dry Ice Blog Post:
Different Phases Of Matter:
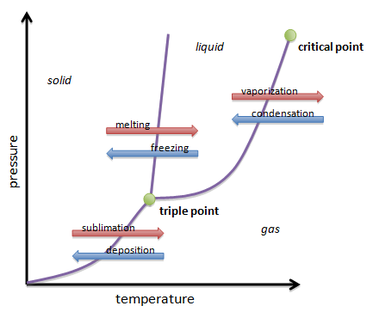
liquid to solid = freezing
liquid to gas = evaporation
gas to liquid = condensation
Solid to liquid = melting
solid to gas = sublimation
gas to solid = deposition
Phase change is the what happens when one matter transforms into another. The three phases of matter are solid, liquid, and gas. The only way to change the phase of matter is through temperature and pressure.
That is all visible through the phase change diagram, which shows the different types of changes.
Liquid is one of the main phases. The molecules that make up a liquid are touching but can glide past each other giving it volume but the ability to form any shape you can image, the most common example of that is pouring water into different sized cups that you may place it in. When a liquid gets colder, it turns into a solid.
A solid is also a main phase. The molecules that make up a solid are compact and are immobile. Which results in it having its own volume.
When a solid gets heated up to a certain extent, it turns into a gas. A gas is the last main phase on the diagram. Gas molecules all all over the place and don't stick together. Therefor it has no volume.
liquid to solid = freezing
liquid to gas = evaporation
gas to liquid = condensation
Solid to liquid = melting
solid to gas = sublimation
gas to solid = deposition
Phase change is the what happens when one matter transforms into another. The three phases of matter are solid, liquid, and gas. The only way to change the phase of matter is through temperature and pressure.
That is all visible through the phase change diagram, which shows the different types of changes.
Liquid is one of the main phases. The molecules that make up a liquid are touching but can glide past each other giving it volume but the ability to form any shape you can image, the most common example of that is pouring water into different sized cups that you may place it in. When a liquid gets colder, it turns into a solid.
A solid is also a main phase. The molecules that make up a solid are compact and are immobile. Which results in it having its own volume.
When a solid gets heated up to a certain extent, it turns into a gas. A gas is the last main phase on the diagram. Gas molecules all all over the place and don't stick together. Therefor it has no volume.
"Properties Of Matter" Blog Post:
Phases of matter is dictated by temp.
Higher energy = Faster moving molecules
Lower energy= Slower moving molecules
Pressure is the sum of the force of the molecules bouncing off of each other.
Temperature affects pressure, pressure changes volume.
Density=Mass/Volume
Increase volume, density goes down, decrease mass, density goes down.
Floating candle, warm up in order to float.
Negative, neutral & positive buoyancy.
In class we did an experiment by created floating candles. Here's how it worked.
We used a plastic bag to create a container for the molecules to expand and grow in. After that we lit the candles underneath the bag. What that candles did was release oxygen from the flame to create energy into our container. As that happened the temperature began heating up the molecules already enclosed in our bag caused them to grow at a fast pace. That makes the mass bigger, but causes the weight of the molecules to deplete and burn away. Essentially, it grows in size, but becomes less heavy, and begins its slow ascension into the air.
Higher energy = Faster moving molecules
Lower energy= Slower moving molecules
Pressure is the sum of the force of the molecules bouncing off of each other.
Temperature affects pressure, pressure changes volume.
Density=Mass/Volume
Increase volume, density goes down, decrease mass, density goes down.
Floating candle, warm up in order to float.
Negative, neutral & positive buoyancy.
In class we did an experiment by created floating candles. Here's how it worked.
We used a plastic bag to create a container for the molecules to expand and grow in. After that we lit the candles underneath the bag. What that candles did was release oxygen from the flame to create energy into our container. As that happened the temperature began heating up the molecules already enclosed in our bag caused them to grow at a fast pace. That makes the mass bigger, but causes the weight of the molecules to deplete and burn away. Essentially, it grows in size, but becomes less heavy, and begins its slow ascension into the air.
Phases Of Matter Pt. 2:
|
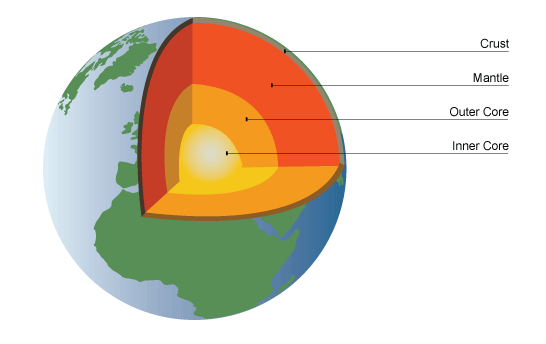
The Earth's crust is made 90% of solid silicates. It's also consist of many different types of igneous, metamorphic, and sedimentary rocks.
The Mantle is made of thick liquid and molten silicates. Silicates have negative charge to them. ( Sio3-2) The Outer Core is made out of liquid iron and nickle, as well as small amounts of other substances. Such as dense elements like gold, platinum, and uranium. The Inner Core is made up of iron and nickle. |
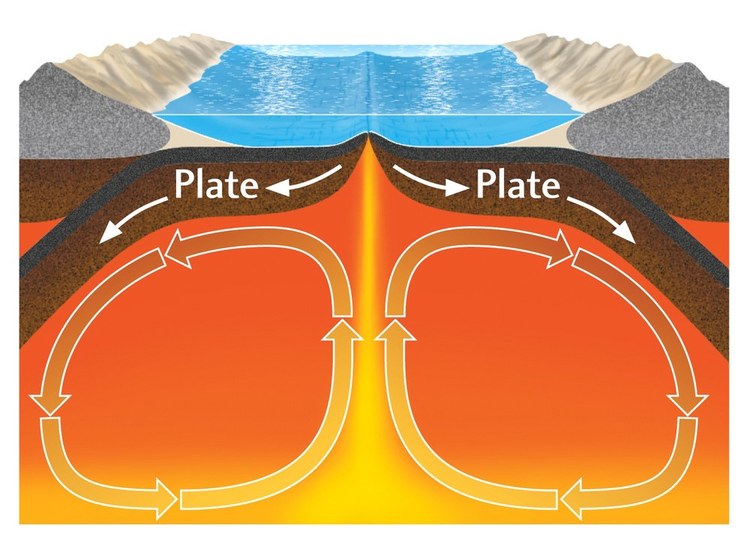
Convection Currents:
Large convection currents in the aesthenosphere transfer heat to the surface, where plumes of less dense magma break apart the plates at the spreading centers, creating divergent plate boundaries.
As the plates move away from the spreading centers, they cool, and the higher density basalt rocks that make up ocean crust get consumed at the ocean trenches/subduction zones. The crust is recycled back into the aesthenosphere.
As the plates move away from the spreading centers, they cool, and the higher density basalt rocks that make up ocean crust get consumed at the ocean trenches/subduction zones. The crust is recycled back into the aesthenosphere.
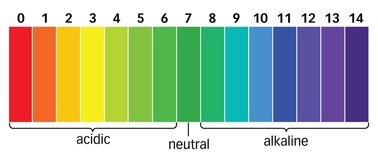
Ocean acidification:
Hypothesis:
I think it's about the acid levels inside of the ocean. Sounds like it's self explanatory.
I think it's about the acid levels inside of the ocean. Sounds like it's self explanatory.
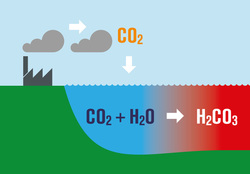
How It Actually Works:
All the water in the world rushes down to the center of the ocean due to gravity. It also brings pollution with it.
The Ocean is absorbing C02 from the atmosphere. The absorbed C02 breaks down to form carbonic acid and hydrogen ions. Free hydrogen ions bond with the available carbonate ions to form bicarbonates, therefor reducing the concentration of carbonate ions. Carbonate ions are required by marine life to build and maintain calcium based structures such as seashells and coral. So it's important not to pollute the air because it even ends up in our coral reefs!
All the water in the world rushes down to the center of the ocean due to gravity. It also brings pollution with it.
The Ocean is absorbing C02 from the atmosphere. The absorbed C02 breaks down to form carbonic acid and hydrogen ions. Free hydrogen ions bond with the available carbonate ions to form bicarbonates, therefor reducing the concentration of carbonate ions. Carbonate ions are required by marine life to build and maintain calcium based structures such as seashells and coral. So it's important not to pollute the air because it even ends up in our coral reefs!
Disappearing Ink Lab:
We did this experiment to demonstrate ocean acidification first hand. Ocean acidification happens when carbon dioxide gets into the ocean and creates a reaction that makes the ocean more acidic. This happens every time carbon dioxide goes into water.
Materials list:
- Distilled water
- Phenolphthalein
- Ethonal Alcohol
- Naoh
- Measure 1g Phenolphthalein into a 125ml Erlenmeyer flask
- Rinse the flask with distilled water
- Fill pipets with 10 milemeter of Ethanol
- Add 100ml D1 H20
- Add (dropwise) 3m Naoh until dark red.